Your Partner in Adipose Stem Cell Technology
Licensed Laboratories & Partnerships
Clinical Partnering
American CryoStem has developed a clinical partnering program open to clinical investigators for use of its ATCELL autologous cellular therapy products. The program provides for reliance on our approved clinical study’s Drug Master File for the investigation of additional therapy options for diseases and injuries with high unmet medical needs. The Clinical Partnering program is available to all international licensees for the extension of our approved US studies under the The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines and can be made available to clinician run and traditional clinical studies. The Company is currently engaged with several partners to extend our clinical studies into orthopedic treatments, wound healing, diabetes, and Duchene Muscular Dystrophy among others.
For additional information please contact:
John Arnone, CEO – Jarnone@americancryostem.com or Anthony Dudzinski, COO tdudzinski@americancryostem.com
International Licensing Programs
Global partners can quickly take advantage of “turn-key” licensing programs that includes completed laboratory processing protocols, marketing materials and developed scientific products to collect process and store individual stem cell samples for clients with the confidence that they are providing cutting-edge science and services. Core to the platform is the collection, processing, storage and laboratory methodologies contained in our proprietary Standard Operating Procedures (SOPs).
Our licensing program can be broken down into four major categories;
(I) Whole adipose tissue collection, processing, storage and return delivery
(II) Stromal vascular fraction processing, storage and return delivery
(III) Mesenchymal cell processing, expansion, storage and return delivery
(IV) Mesenchymal cell culturing, conditioned media creation and storage
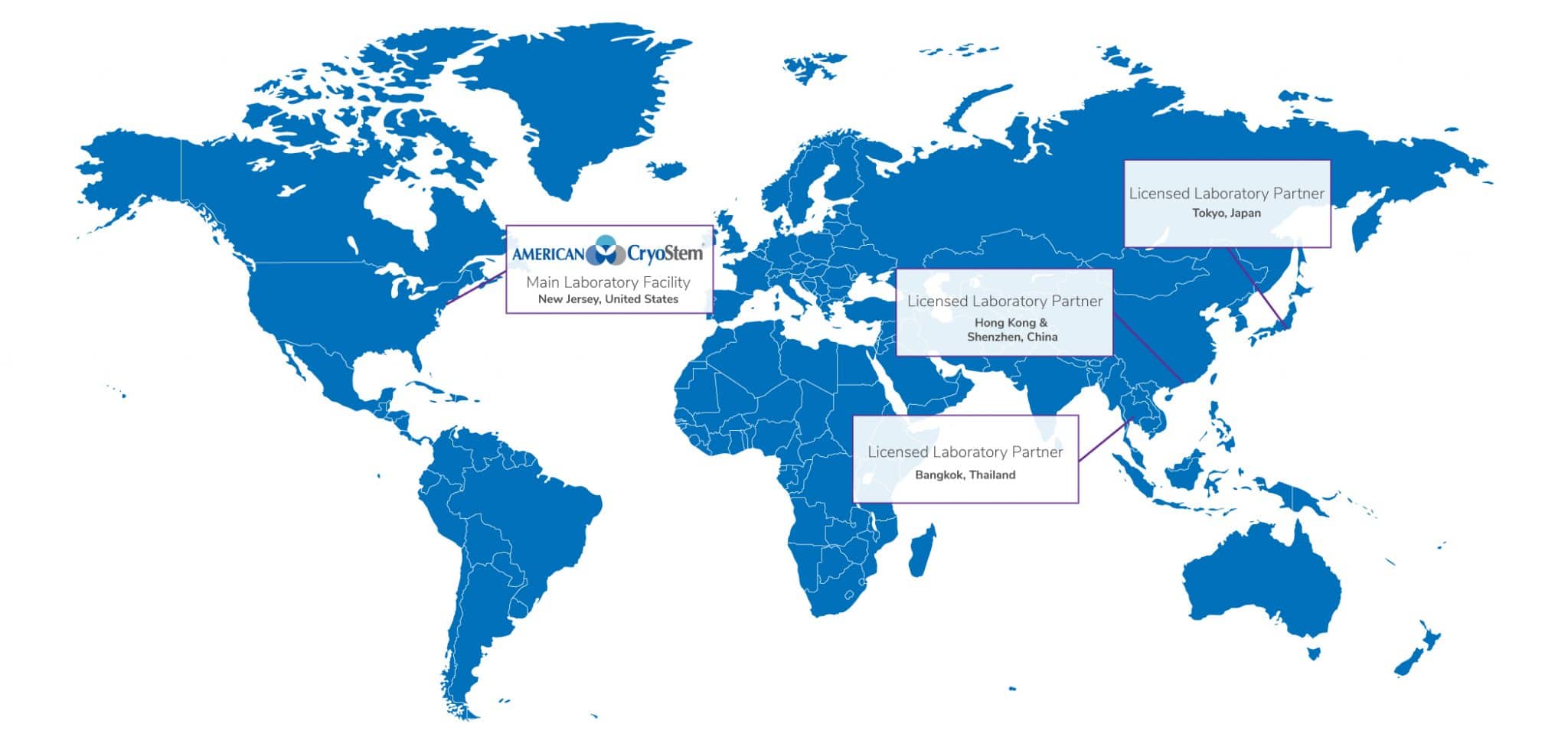
Our Licensed Facilities
American CryoStem continues to expand its international licensing program into emerging Regenerative and Personalized Medicine markets. We are committed to bringing our first in class adipose tissue based stem cell platform into the forefront of the industry as our partners work within their regulatory framework to provide tissue and adult stem cell based services to their clients.
Baoxin Asia Pacific Biotechnology (Shenzhen) Co. Ltd.
Baoxin Asia Pacific is an innovative cryopreservation technology company based in Shenzhen China, dedicated to exploring and developing innovative biotechnology and business innovation models. The Company operates state of the art processing and storage facilities in Schenzhen and at the Futian International Bio-Pharmaceutical Industrial Park Baoxin’s research and development strategies include stem cell therapeutics developed in-house in Chinese laboratories, and biotechnologies introduced from partners for potential cosmetic, aging and chronic diseases in China and Asia Pacific. To improve our research and development capabilities, Baoxin Asia Pacific is building a strong integrated R&D platform from laboratory to clinical trials that include its licensed technology for American CryoStem Corporation, to meet the wide-ranging needs of people's regenerative medical and regenerative medical markets in China and throughout Asia
Health Innovative Technology Corporation Limited
Health Innovative Technology Corporation Limited (HIT) (www.healthinnotech.com) is a healthcare platform dedicated to safeguard the health of people in Greater China by introducing various innovative and scientifically proven biomedical technologies from around the world, including stem cell storage and genome diagnostic services. HIT strives to promote global development of life science and medical technology by supporting research projects around the globe. HIT imports cutting-edge life science technologies that may contribute to health prevention.
Cryoviva (Thailand) Ltd.
Cryoviva (Thailand) Ltd. was established in 2007 with the cooperation of leading companies in the world such as Indorama Ventures Public Company Limited Thailand, Cryoviva Biotech Pvt. Ltd. (formerly known as Cryobanks International India), and RJ Corp India. Cryoviva is part of a large group engaged in healthcare comprising of stem cell banks, stem cell expansion facilities, diagnostic labs and centres and a maternal hospital. The Company has been certified by AABB and the ISO 9001: 2008 Quality Management System. Cryoviva (Thailand) Co., Ltd. is the only stem cell bank that has been approved by the Board of Investment of Thailand (BOI) and accredited by AABB in Thailand and awarded Frost & Sullivan’s Thailand Stem Cell Company of the year for three consecutive years between 2015-2017.
